Dr. Michael Greger, of NutritionFacts.org, gave a talk a while back which was posted to YouTube in 2013. It’s much more substantial than the sound bite videos he shares on his site, but tackles a substantial topic.
Dr. Greger asks: If vegetarianism and veganism improve biomarkers for health, then why haven’t studies shown an effect on mortality for vegetarians and vegans vs. omnivores? Literature reviews released after this video was recorded (e.g. 2015, 2017) continue to report similar observations: they saw reduced risk from ischemic heart disease (aka atherosclerosis and heart attacks) and reduced risk of cancer, but they did not find reduced risk of total cardiovascular disease (e.g. stroke, heart failure) or total mortality. Why would there be no difference in total mortality — in other words, why don’t the vegetarians and vegans seem to be living longer? It’s like, the diet looks great on paper, but the benefits don’t cash out.
The talk advances two main hypotheses of what may be lacking in the veg*n diet compared to omnivore. His primary hypothesis, which I want to focus on in this post, concerns omega-3 fatty acid intake, or ω-3. The secondary hypothesis is that veg*ns might have elevated homocysteine or neurological issues because of suboptimal vitamin B12 intake. Regarding B12, deficiency is easy to test for and easy to fix with a supplement, so you should just supplement B12, in fact you probably should do that even if you are an omnivore.
Background
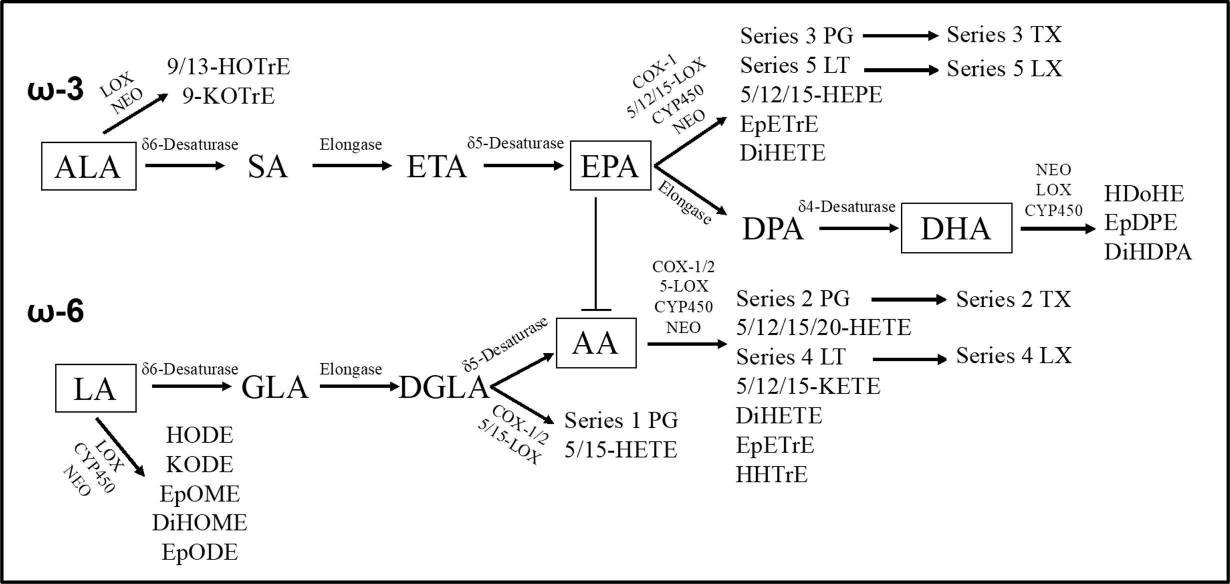
Let’s talk about polyunsaturated fatty acids (PUFA) then. For the basics, you can read up on Wikipedia but I made a diagram to spice up the post.
Both ω-6 and ω-3 are essential in the human diet, though in terms of absolute requirement, only tiny amounts are needed. The most abundant source of ω-6 is a short-chain fatty acid called linoleic acid (LA), and for ω-3 it’s alpha-linolenic acid (ALA), both from plants. The body may require forms of ω-3 or ω-6 with longer carbon chains for some of its functions, but it has mechanisms to elongate the molecules. It’s debatable how well those mechanisms work, but the point is that there is a way for the body to modify chain length, while it doesn’t have a way to modify the straightness of the chains, so the two PUFA types have to be obtained as raw materials.
If you are a veg*n, you may wind up consuming a lot of nuts and seeds. That means a lot of PUFA, and if you are doing something like vegan keto, ω-6 LA may become a main source of calories. Is that good or bad? It’s certainly novel, nutritionally speaking. At the beginning of the 1900s, most of the fats consumed in the USA were animal based (lard and butter), so mostly saturated and monounsaturated. Around that time, processes were invented to extract oil from seeds that are not traditional sources (e.g., corn, cottonseed) and thereby make use of industrial waste products. Then, scientists started denouncing animal-derived saturated fats and this created a market for all those cheap new oils. So heavy use of ω-6 LA is now widespread, but we don’t have enough history with it to know if there are long-term health impacts. Researcher Stephan Guyenet observed that we are “living an uncontrolled experiment in the modern world” as demonstrated by such effects as LA content of human subcutaneous body fat increasing from ~10% to >20% from 1960 to 2010 in the US.
Unlike ω-6, ω-3 is pretty hard to come by from vegetable sources. Most nuts and seeds have much more LA than ALA (see below graphic). The notable exceptions are flax and chia. People also cite walnuts. So in his talk, Dr. Greger calls out that studies rate omnivores as consuming ω-6 and ω-3 in a 7 to 1 ratio, vegetarians a 10 to 1 ratio, and vegans 15 to 1 or more (one study found 44:1). So the combination of abundant ω-6 and scarce ω-3 in plant foods may naturally lead to a poorer ratio in those getting most or all of their calories from plants. Dr. Greger argues that improving this ratio can decrease the production of a long-chain ω-6 called arachidonic acid (AA) which supposedly causes inflammation. Hooray, we can unlock the mortality benefit that the veg*n diet promises!
I decided to look into this myself, since I currently ingest rather a lot of ω-6 LA and I wanted to have reasonable confidence that it’s safe. What follows are my findings, and of course note that I am not a health professional and you should use this information at your own risk.
Is increased ω-6 LA intake harmful?
Influence on AA levels
The main concern about ω-6 intake seems to be the fact that LA is a precursor to a long-chain fatty acid called arachidonic acid (ARA or AA), and AA in turn is a precursor to a number of pro-inflammatory substances. In Dr. Greger’s talk, he states that AA “goes on to produce inflammation,” perhaps implying that a lower absolute amount of AA might be desirable. However, the real situation seems to be more nuanced. Consuming AA directly doesn’t seem to increase concentrations of inflammatory markers.[1] A 2011 review found that increases and decreases in LA intake had no effect on tissue AA levels.[2] So, even though there’s no dispute that LA is a precursor to AA, there isn’t obvious evidence that increasing intake of LA will necessarily lead to excessive or uncontrolled levels of AA or related inflammation.
In Peter Attia’s Dec 2019 podcast with lipid expert Bill Harris Ph.D., Dr. Harris confirms that there is practically no correlation between AA levels and LA intake, because AA levels are tightly controlled in the body and less than 1% of consumed LA is made into AA. He says: “At the end of the day if ω-6 levels are high and that’s associated with reduced risk for diabetes and heart disease, that’s a very powerful testimony to me that the ω-6s are good.”
By the way, it’s worth calling out that AA isn’t bad just because it’s “pro-inflammatory.” AA is the most predominant long-chain polyunsaturated fatty acid in human milk,[3] and inflammatory processes aren’t necessarily bad. It’s just when inflammation goes on chronically that it’s a problem.
The oxidation theory
Another argument I have seen is that ω-6 PUFA is bad because it is prone to oxidation. It is true that PUFAs spoil much faster than monounsaturated or saturated fats. This general argument comes in two flavors. In the first, the concern is that ω-6 LA may be oxidized at the time that you eat it. This post by a Paleo diet proponent is an example of this argument. I’m not convinced by this claim, given that ω-3 is even more vulnerable to oxidation than ω-6, and I have yet to see anyone argue that you should avoid eating ω-3 because it might be oxidized. Anyway, don’t eat rancid oil! Note that if your oil is sticky or tastes bitter, it may be rancid. This is not something mysterious or sneaky, except perhaps if you are taking pills (in which case you should probably cut one open and taste the contents). I find that flaxseed oil, which is about 50% ω-3, normally has a mild taste but it turns bitter if I leave an open bottle in the refrigerator for more than a month or so. I’ve observed the same thing in the case of whole grain wheat flour, which contains the ω-6-rich germ. Store-bought whole wheat flour, in my experience, has a bitter note, but I never realized this or recognized it as a defect until I tried grinding my own whole-wheat flour and noticed that it was free of the bitterness. Discovering this was the moment when I came to understand why white refined flour has become dominant: the longer shelf life. Anyway, for sure, don’t eat rancid oils.
According to the second form of this argument, LA gets incorporated into LDL cholesterol, and once on board the LDL, the LA is prone to cause the LDL to oxidize, and oxidized LDL leads to atherosclerosis. Apparently it’s not controversial that oxidized LDL is involved in atherosclerosis — and this makes sense intuitively, because oxidized PUFAs harden; oil paint is nothing but PUFA — but the theory of causation seems to be speculative. I only found two references for this theory on my safari through PubMed.[4] Although I didn’t spend a ton of time looking into this, I am skeptical for two reasons. First, the theory is positioned as an answer to the question of why oxidized LDL exists to be part of the atherosclerotic process. The proposed answer: because there is too much LA in the LDL, which makes it vulnerable. I think this begs the question though. There already was a lot of LA in the LDL (“Linoleic acid is the most abundant fatty acid in human low density lipoproteins (LDL)”[5]) so the real question in my mind is: what is causing the LA in the LDL to get oxidized? Clearly this process is happening more aggressively in atherosclerotic patients than healthy patients, and it is not clear to me why increasing LA in LDL is inherently causative. Second, if the effect is meaningful, then the studies on the outcomes of replacing saturated fat with ω-6 (discussed in next section) should have been showing negative effects on mortality instead of neutral.
Evidence of impact on overall mortality
Harvard Medical School released a 2009 statement (updated in 2019), “No need to avoid healthy omega-6 fats,” in which it dismissed the concern that ω-6 intake may cause inflammation: “The AHA reviewers found that eating more omega-6 fats didn’t rev up inflammation. Instead, eating more omega-6 fats either reduced markers of inflammation or left them unchanged. Many studies showed that rates of heart disease went down as consumption of omega-6 fats went up.” The statement referred to several sources but didn’t explicitly name most of them; the only one I was able to track down was the American Heart Association review which declared that “At present, little direct evidence supports a net proinflammatory, proatherogenic effect of LA in humans.”
I also found a number of studies and reviews which mostly indicated no benefit from ω-6 intake on mortality or cardiovascular disease. In this 2016 re-evaluation of the Minnesota Coronary Experiment, replacing saturated fat with ω-6 LA decreased serum cholesterol but this did not translate to a lower risk of death. In this 2018 Cochrane review, there was little or no difference to all-cause mortality or cardiovascular disease. In findings published in 2013 from the Sydney Diet Heart Study, replacing saturated fat with ω-6 LA without adding ω-3 increased all-cause mortality and cardiovascular disease, but I wasn’t impressed because this study was conducted from 1966-73, before we were hip to trans fats, and the study permitted margarine. On the whole, I didn’t find any evidence of harm from ω-6 intake.
Safety of very high intakes of ω-6
The AHA review notes that intakes above 25% of energy may be uncharted territory. I eat about 1600 calories/day so that means I should stay under 400 calories from LA. At 9 calories per gram, that’s 44 grams. I have a couple of recipes that get to the high 30s for a day’s worth, for example pesto based on pine nuts, but it doesn’t seem hard to stay under that limit.
Does ω-6/ω-3 ratio matter?
ω-6/ω-3 ratio vs. AA/EPA ratio
According to Dr. Greger’s talk, intake of ω-3 ALA decreases inflammation by competing with ω-6 LA for access to an enzyme. This bottleneck enzyme converts ω-6 LA to AA (which is inflammatory) and converts ω-3 ALA to a “magical substance” called EPA (which is anti-inflammatory). If we are getting too much ω-6 LA and/or not enough ω-3 ALA, then — the argument goes — the enzyme will make too much AA and not enough EPA.
The idea that LA and ALA compete for the same set of enzymes seems to be well accepted in the literature, as I saw it referenced in papers from 2012[6] through 2019.[7] But, now we know that too much AA is not a concern. So, does a poor ω-6/ω-3 ratio depress EPA even if AA does not elevate? In other words, does LA/ALA ratio influence AA/EPA ratio?
For what it’s worth, the ω-6/ω-3 ratio seems to be falling out of favor. In the Attia podcast, Dr. Harris disparages the ratio measure, saying “I think it doesn’t make any sense, partly because it presumes that the ω-6s actually are bad, and they really aren’t.” The ratio has been criticized because the same ratio number can describe a condition where both ω-6 and ω-3 are deficient or sub-optimal, and where both are replete. This measure also doesn’t distinguish between short-chain and long-chain forms, which have different effects in the body.[8] And I guess people were disappointed by the outcome of the OPTILIP study which was supposed to show benefit on cardiovascular disease as a result of decreasing the ratio, but didn’t.
So let’s look into whether ω-6 LA intake spells trouble for EPA levels.
Effect of ω-6 LA intake on EPA levels
I found a study from 2010 in which decreasing the LA:ALA ratio from 10:1 to 2:1 increased EPA by 60%. But I didn’t have access to the full text and without reading that it wasn’t clear if the LA intake was kept constant between the two treatments. So the results could be explained by the decrease in LA, by the increase in ALA, or by the change in ratio.
In this 2005 study, men were given supplements of either flax oil (1:2 ω-6/ω-3 ratio) or sunflower oil (28:1 ω-6/ω-3 ratio). The 1:2 diet obtained 7% of calories from ALA and 3% of calories from LA, while the 28:1 diet obtained 0.5% of calories from ALA and 9.3% from LA. There was also a control group holding LA and ALA constant. In the 1:2 diet, EPA increased >2-fold over baseline. This seems to demonstrate that increasing absolute intake of ω-3 ALA while holding ω-6 LA constant can increase EPA conversion.
In this 2007 study, decreasing ω-6 LA while keeping ω-3 ALA constant (going from 10:1 to 4:1 ratio) also increased EPA. The 10:1 diet had 10.5% energy from LA and 1.1% energy from ALA, and the 4:1 diet had 3.8% energy from LA and 1.0% energy from ALA. So the competition phenomenon seems to be real, though though here the absolute intakes of ALA were lower (1% of energy instead of 3%) so it’s possible there is a threshold under which scarcity engages the enzyme competition mechanism. Doesn’t seem likely, just sayin.’
A 2018 study found that supplementing a tablespoon of ω-3 ALA per day increased ALA concentrations in red blood cells and also increased EPA, though it decreased DHA by the same amount as EPA increased.
I decided to overlook this 2006 study which claims to find that conversion outcome is not driven by ratio. The study design is great — the control group was given PUFAs in a 19:1 ratio, and two experimental groups were each given a 7:1 ratio, one by lowering LA and the other by increasing ALA — but (1) I don’t understand why the high-ALA group had a much greater incorporation into ALA compartment during the run-in period (111 mg vs. 72-74 mg in low-LA and control respectively) when all three of the groups were supposed to be on control diet during the run-in period. Doesn’t this call the result into question? And (2) the results found that EPA conversion was greatest in the low-LA group, and in particular the group given added ALA didn’t see increased EPA conversion at all, which seems counterintuitive and contradicts the findings from the other studies we just looked at. Moving on.
So the picture is still hazy but it’s consistent with the view that ω-6/ω-3 ratio does influence the AA/EPA ratio.
Can a favorable AA/EPA ratio be obtained without supplementing EPA?
There are some widely held theories about what a good ω-6/ω-3 ratio is (4:1 to 1:1 or so), but what we are really interested in is getting the right AA/EPA ratio, so do we know what the target is?
This 2019 review states that “An AA/EPA ratio between 1.5 and 3 has been suggested as the optimal range in order to reduce cellular inflammation and achieve beneficial effects in different clinical settings….” This 2013 review says of a recent study that “A significant reduction in the risk of coronary events was observed when the ratio of EPA to arachidonic acid (AA) (EPA/AA) was > 0.75.” (Expressed as AA/EPA, that target would be < 1.3).
The next question, then, is whether a favorable AA/EPA ratio can be obtained without supplementation of pre-formed EPA, i.e. through manipulating intake of LA and ALA alone. If we want an AA/EPA of 1.5-3, can we hit that?
In the 2005 study, when they implemented a diet with twice as many calories from ω-3 ALA as ω-6 LA, they saw a decrease in AA/EPA from 13.9 to 5.5. That’s still not as low as we want it to be, and if you happen to be following a diet high in ω-6 LA, you would have to basically mainline flax oil to get double the amount of ALA.
The evidence here is thin, but it’s pointing to the conclusion that you may not be able to get an “optimal” AA/EPA ratio from consuming ALA alone. If an ideal AA/EPA ratio is important, the most prudent thing to do is supplement with pre-formed EPA. If you are unable or unwilling to supplement, does it make sense to lower your ω-6/ω-3 ratio in an effort to improve AA/EPA even if you don’t get to “optimal”? Unclear. There is reason to doubt since (a) increasing ω-6 intake doesn’t seem to cause harm and (b) studies like OPTILIP (reducing the LA/ALA ratio from 10 to 3-5) didn’t show benefit on biomarkers from that change.
Safety of very high intakes of ω-3
This may be moot, but if you should want to eat a large amount of ω-3 ALA in order to level out the ratio when you happen to be eating a lot of ω-6 LA (upwards of 20% of calories, say), is this safe? It’s not clear if anyone has studied the effects of ingesting massive quantities of flax oil. In this study, the experimental diet provided 6.5% energy from ALA and that’s about the most I could find; 15%+ of energy from ALA might be a human nutritional frontier.
Almost all the evidence on safety that I could dig up concerned fish oil consumption. The big concern is that excessive ω-3 intake may increase bleeding. Dr. Weil’s article on the topic says “Very large intakes of fish oil/omega-3 fatty acids (termed ‘Eskimo amounts’ by the National Institutes of Health, referring to diets that consist almost exclusively of fish) may increase the risk of hemorrhagic stroke, and have been associated with nosebleed and blood in the urine.” According to This 2006 review, “Eskimo amounts” would be 7g/day of long-chain ω-3; this paper also says that the concern about bleeding arises because the Greenland Inuit consuming this diet may have had a relatively higher prevlalence of cerebral haemorrhage, however I get the impression the concern is bolstered by observations that ω-3 PUFAs can have an anti-thrombotic effect[9] (i.e., can inhibit clotting — which is considered good if you are at risk of blood vessel blockage as part of a disease process, but bad if you have healthy blood vessels that rupture and then can’t repair fast enough). Reportedly, the FDA recognizes 3g/day EPA plus DHA as safe[10] though larger doses have been used for therapeutic purposes, as suggested by this popular press article describing the use of up to 20g of fish oil a day in recovery of traumatic brain injury. In 1995, doses of 4g of fish oil per day did not increase bleeding. But still, this is all fish oil, not ALA.
I didn’t find much about ALA specifically, but what I did find was reassuring. In a 2010 review the authors state, “flaxseed ingestion does not influence platelet function in healthy individuals. One might expect flaxseed and its content of ALA to induce an inhibition of platelet aggregation because fish oils (and EPA and DHA) are well-known inhibitors of platelet aggregation. However, many studies have not detected any changes in platelet aggregation after supplementing the diet with flaxseed for one to three months” although “Only two studies have shown some changes in platelet aggregation as a function of flaxseed ingestion; in one of these, the changes were isolated and modest at best. More extensive dose-response studies are needed to assess the association between ALA and any changes in bleeding times before definitive conclusions can be drawn.” Another review, from 1997, states more confidently that “Only long-chain n-3 fatty acids prolong the template bleeding time.”
There is also my personal experience. As of this writing (mid-2020) I have been following a diet with >20g of ALA per day for maybe a year, and I haven’t seen increased nosebleeds or anything like that. So I will assume it’s not a risk worth worrying about — but again, fellow biohacker, use this information at your own risk.
Is it important to supplement DHA?
DHA has kind of a mystical status among ω-3s because it is uniquely abundant in eye and brain tissue. Other than cannibalism, sources of this fatty acid are mainly human breast milk, cold water fish, and marine algae (the algae is mostly where the fish get it from). Here are some reviews from 1999 and 2016 highlighting the importance of DHA in human development and possible therapeutic opportunities like depression, cardiovascular disease, and dementia.
The human body can create DHA out of EPA, but one thing that I saw in study after study was that you don’t get measurable increases in DHA from consumption of plant-based ω-3 ALA. In theory, if you can raise EPA by increasing ALA intake, then with more EPA around as a substrate, you should wind up with increased DHA too, right? Well, the problem is, conversion of EPA to DHA requires Delta 6 Desaturase (D6D) which is an enzyme that is also required to convert ALA to EPA. So this sets up a catch-22 in that the more ALA you eat, the more you dilute the substrate pool for D6D and compete with the small amount of additional EPA that is being created. The effect is explained in this paper. The picture is probably even worse if you happen to be consuming a lot of ω-6 LA because if you take another look at the illustration in Figure 3, D6D also appears in the LA to AA conversion process[11] so the LA is going to dilute the total substrate for that enzyme even more. In fact, according to this review, even directly supplementing DHA’s precursor, EPA, doesn’t seem to have much of an impact. It looks like, if boosting DHA is important, it needs to be supplemented directly.
Wait, what about long-term veg*ns? We know about cases where die-hard 20+ year vegans developed neurological problems from B12 deficiency. Have we seen similar issues from DHA deprivation? Fortunately, someone looked into this in 2005. “Plasma EPA, DPA, and DHA proportions were not significantly associated with the duration of time since the subjects became vegetarian or vegan, which ranged from <1 y to >20 y. … This finding suggests that when animal foods are wholly excluded from the diet, the endogenous production of EPA and DHA results in low but stable plasma concentrations of these fatty acids.” This 2009 review concludes, “There is no evidence of adverse effects on health or cognitive function with lower DHA intake in vegetarians.” So we have reason to believe that the body can keep itself out of trouble here.
The question becomes: can we do better? If we supplement DHA, will we become euphoric geniuses with 20/20 vision? I don’t know, but on the other hand, there’s little harm. DHA supplementation is pretty safe (per earlier discussion of high intakes of ω-3) and doesn’t even require compromising on veg*n lifestyle, as the algal oil supplements seem to be effective.
Personally, my biggest concern with supplementation is about freshness. If one has to choose between taking pricey supplements with partially oxidized EPA and DHA, and not supplementing at all, which is worse? (There is some suggestion that it’s better to take it, even if oxidized.) Or is it worth the trouble and expense to get cold-chain fresh cod liver oil shipped to you? Or, just suck it up and eat fish? (But but, mercury, and one time I saw a worm pop its head out of a raw salmon fillet that I got at a grocery store [shudder]…)
Conclusion
We observed that ω-6/ω-3 (LA/ALA) ratio does influence AA/EPA ratio, and a low AA/EPA ratio is associated with lower inflammation and better cardiovascular outcomes. Yet high ω-6 LA intakes are not associated with bad outcomes, and it was the high LA intake in the vegan diet that we were worried about. Also, it doesn’t look possible to get the AA/EPA ratio into “optimal” range through ALA intake alone (i.e., consuming more flax).
So are vegans at particular risk? I see two main possibilities:
-
The AA/EPA ratio isn’t a big deal, as suggested by (a) the AHA reviews that found no health risk from ω-6 intake, and (b) the stable blood levels in long term vegans. This would mean that if there is any issue, it’s explained by frank deficiency, so it would be enough for vegans to get some ω-3; the ratio doesn’t matter. In this case it seems unlikely that there is a systematic problem among vegans since anyone eating a reasonable amount of soy is probably avoiding ω-3 deficiency.
-
The AA/EPA ratio is important and vegans are at risk unless they supplement pre-formed EPA. In this case, Dr. Greger’s “just the flax, ma’am” advice would not be a strong enough intervention.
Either way, intake ratio for LA and ALA doesn’t seem to matter, so I am not convinced that feeding vegans more ω-3 ALA will solve the “vegan paradox.”
My personal takeaways, after doing this research? (a) Stop worrying about adding flax oil to my recipes, and (b) do an experiment with supplementing EPA and DHA.